Plans
Communications - The Electronic Flame
Dear Editor:
I read Thomas E. Fairbairn's article, "Electronic Flame," in the December, 1952, issue with considerable interest — not in the phenomenon itself but rather in the nature of its manifestation.
The electronic flame described in the subject article is I believe akin to the natural manifestation broadly classified as St. Elmo's Fire, which has always frightened seafaring men. The same phenomenon frequently makes flyers jittery when flying through thunderstorms. Sometimes every projection on the aircraft seems to be on fire.
Under another name, "precipitation static," a lower potential form which does not "flame," it makes radio communication with the aircraft impossible. It is quite common for aircraft flying in certain kinds of weather to accumulate a static charge of more than 250 kilovolts.
Mr. Fairbairn asks, "What is this electronic flame?" I will attempt to answer him:
He states that glass conducts current only when in the melted state. This is not entirely correct. Glass will conduct current, though very slightly, even at ordinary room temperatures and its conductivity progressively increases with increasing temperature. In fact, two electrodes sealed into a glass bead make an excellent temperature-sensitive resistor for use at temperatures above about 300° C. Therefore, in view of the extremely high potential at the end of the tank coil and the small current required by the electronic flame, a sufficient number of electrons can pass through the cold glass rod to form the flame.
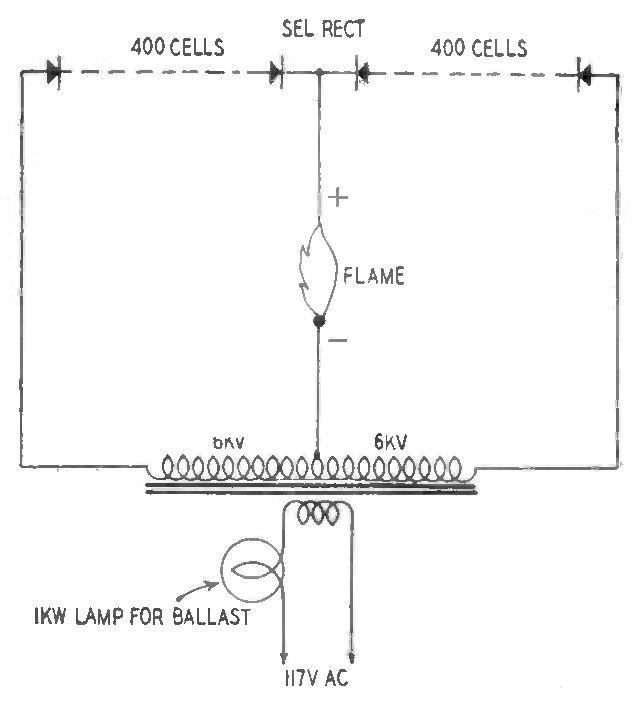
We have an apparatus which does the same job as Mr. Fairbairn's 1-kw, 14-mc transmitter. Ours was built at a very small fraction of his cost; it has extremely modest power requirements and should not offend the FCC. This apparatus was built for electroforming high-voltage selenium rectifiers. The complete circuit is shown in the sketch on page 125. The transformer is an ordinary 12-kv, 24-ma neon sign unit which happened to have the mid-point of the high-voltage winding grounded to the case. It is necessary to refer to this apparatus because the phenomena connected therewith serve to explain the why of the electronic flame.
With this apparatus, the ends of two wires connected to the positive and negative outputs are spaced 1/4 to 1/2-inch apart and the line cord is plugged in. The flame starts promptly.
Here a curious thing happens. The end of the wire which is connected to the negative terminal glows brightly and melts into a ball on the end of the wire. The wire connected to the positive terminal does not melt, and, if of the same diameter as the other wire, hardly reaches a dull red heat. If the wires are of iron, a brilliant shower of sparks (incandescent iron particles) erupts from the negative wire during the melting, which takes place in only a few seconds.
The melting of the negative wire widens the gap, and therefore the length of the flame, to about ¾ inch, at which point no further melting occurs and the flame settles down to a nice silent fire drawing 30-35 ma at 2,000 volts.
The flame is fire in every sense of the word. Inflammable materials introduced into the flame promptly burn. Small-gauge copper wire held in the flame promptly melts. Light emitted by the flame and by the incandescent ball on the end of the negative wire extends from the infra-red to the ultra-violet. Fluorescent materials fluoresce, though weakly, when held near the flame. The spectrum of this flame and the incandescent negative wire has a sharply defined orange line corresponding to a wavelength of approximately 0.6 micron. A piece of glass placed in the flame produces the characteristic sodium line in the spectrum. It is my opinion that the aforesaid orange spectrum line is due to burning of dust particles in the air surrounding the flame, or more probably to one of the rare gases in air.
That this is a flame rather than the customary corona or spark discharge is evidenced by the total absence of the odor of ozone which is characteristic of such discharges. Like any flame, this one wavers about with each small draft of air.
I have found this phenomenon useful for welding copper-iron thermocouples made of up to No. 20 AWG wire or for welding copper wires together. For welding, the flame should be turned off as soon as the molten ball forms, because if the flame is continued the whole ball becomes converted to oxide which is very brittle and may be broken off with the fingers.
This flame seems to be truly an "electronic flame" in every sense of the term. The reason for only the negative wire melting is due to the following actions, two of which occur also in Mr. Fairbairn's high-frequency version:
1. The fact that current does flow through the flame is conclusive proof that a large number of electrons are moving through it. These electrons presumably leave the surface of the negative wire at high speed, which enables them to ionize the gas molecules comprising the air with which they must collide with considerable force. The flame therefore must be composed largely of ionized gas, plus free electrons. As the electrons must be literally torn from their orbits in the metal atoms comprising the negative wire, the energy level of these surface atoms rises. With a rise in energy level, there is an increase in temperature, which tends to free more electrons from the surface atoms. Electrons continue to be torn out of the surface orbits faster than they can be replaced by the relatively slow movement of electrons through the wire until at last the metal comprising the end of the wire melts. In melting, the characteristic ball is formed, presenting a larger surface from which the electron supply can be secured, thereby drastically reducing the energy per unit area and permitting the ball to cool below the melting point.
2. The surface of the negative wire must be simultaneously bombarded by positive gas ions in the portion of the flame adjacent to the negative wire; some heat must be contributed by the collision; the positive ion extracts electrons from the surface atoms.
3. Oxidation is of itself exothermic, once the temperature reaches a critical temperature depending upon the metal. That is, above a certain temperature, the reaction is accompanied by the liberation of heat.
In the high-frequency version of the electronic flame, the melting of the wire or glass must be due to bombardment of the tip by the ionized gas particles comprising the flame, and, in the case of wire, by exothermic oxidation.
H. B. CONANT
Conant Laboratories
Lincoln, Nebraska